In the Spring of 2020, at the height of the pandemic, BML Technology launched the COVID-19 Emergency Digital Health Task Force encompassing professionals from within the Montreal digital health ecosystem. At the time we did not have a clear mission, but we understood that at this moment of crisis we could help our healthcare systems, […]
Ryerson University’s MedTech Talent Accelerator, in association with McGill University’s Biological & Biomedical Engineering (BBME) Student Society, recently hosted its virtual 2-day Biomedical Engineering symposium on May 5-6, 2021. Its main goal was to provide students with an opportunity to showcase their work to peers, faculty, and invited guests. As part of the Symposium a virtual […]
Ryerson University’s MedTech Talent Accelerator, in association with McGill University’s Biological & Biomedical Engineering (BBME) Student Society, will be hosting a virtual 2-day Biomedical Engineering symposium on May 5-6, 2021. As part of the program, a virtual panel discussion entitled ‘Explore Medtech: Combating COVID-19’ will be held May 6th, 2021 at 2:30 PM EDT – […]
The Montreal based, COVID-19 Digital Health Task Force held its most recent quarterly meeting on February 26th, 2021. The Mission of the Task Force is to help raise the level of COVID-19 related digital health projects in the ecosystem and to guide them with their resources, experience and community to ensure they can adequately meet a real […]
The last meeting of the Montreal-based COVID-19 Emergency Digital Health Task Force took place on February 26th, 2021. The topic of discussion was the potential utility of COVID-19 rapid testing and tracing in Canada, an important topic considering the impact COVID-19 has had on our citizens and our economic and social lives. And the knowledge […]
Since the beginning of the COVID-19 pandemic in Canada, myriad efforts and strategies have been explored to slow the spread of the virus and contain the damage it causes. The primary strategy deployed has been a series of lockdowns and restrictions on social and economic activity. The intention of these lockdowns is to ‘flatten the […]
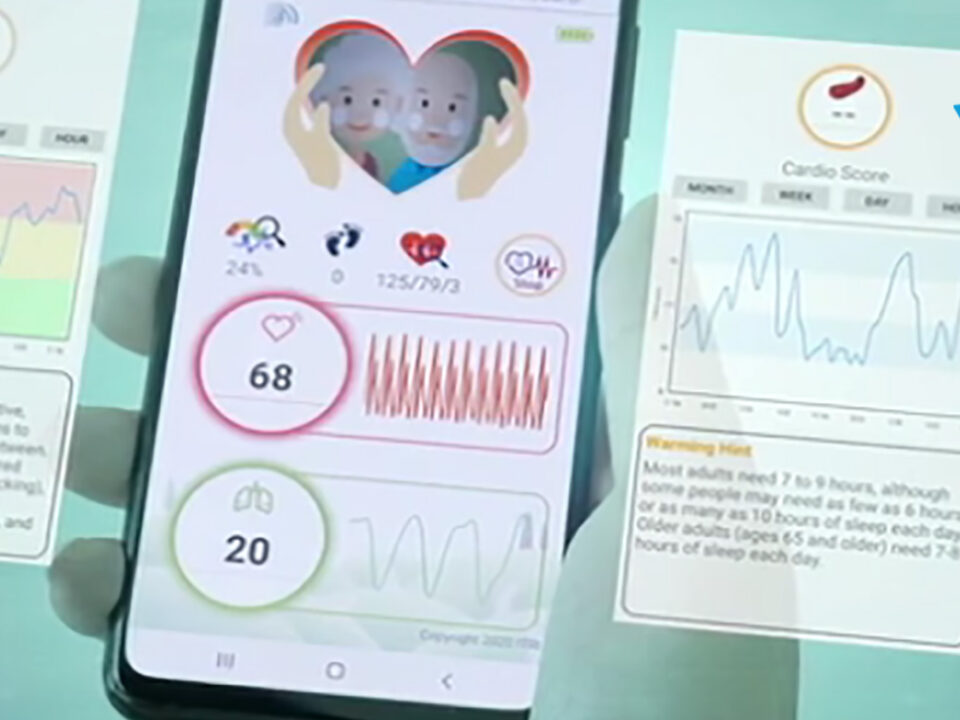
CES is the premiere event where new consumer technology and innovation are revealed to the world. It is a proving ground for breakthrough technologies and global innovators and showcases companies in consumer technology hardware, content, technology delivery and more. One of the sectors demonstrating the greatest innovation and growth in tools and technologies empowering consumers […]
RRP Canada Rapid Response Platform has relaunched its website that connects Canadian manufacturers and suppliers of PPE (Protective Personal Equipment) with the companies, institutions and other organizations that require it. The Rapid Response Platform site has evolved into one of Canada’s most trusted sources of PPE. The project was funded by Industry Science & Economic […]
In the early days of the COVID-19 pandemic, and already with the insight that our health care system would be pushed to the limit with regards to the availability of COVD-19 related medical devices, PPE and specifically ventilators The Montreal General Hospital Foundation and the Research Institute of the McGill University Health Center (IR-MUHC), in […]
The COVID-19 Personal Protective Equipment (PPE) Supply Crisis in Canada The COVID-19 pandemic has created an unprecedented need for Canadian solutions to medical devices, materials and equipment. Specifically in the area of PPE the needs are substantial and traditional supply chains have been overwhelmed by local and international demand where in many cases countries hoarded […]
There is an excellent “Women in Health Technology” virtual conference event showcasing women founders and entrepreneurs in health technology to be held on December 10th, 2020 at 5PM. The National Research Council Canada / Conseil national de recherches Canada, Hacking Health Global and Prompt will be hosting Josette-Renee Landry of Streamline Genomics, Jade Doucet-Martineau of […]
The BML COVID-19 Task Force is offering a free, practical workshop on “Decoding Health Canada’s accelerated access programs” with real world case studies. Join us on Friday August 7 at 1 pm EST. Spaces are limited. Register here now. Participants will: Understand specifics of Establishment License (MDEL) and Clinical Evaluation (ITA) applications Learn Interim Order […]